The role of laboratory test biomarkers in diagnosis, risk assessment, and monitoring of COVID-19 patients
DOI:
https://doi.org/10.5281/zenodo.7559225Keywords:
Biomarkers, COVID-19, diagnosis, prognosis, SARS-CoV-2Abstract
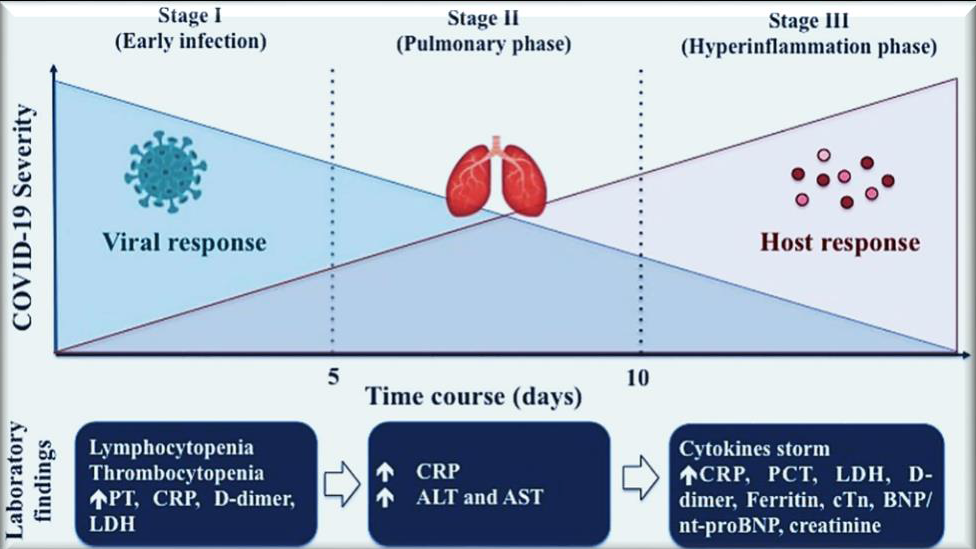
Government officials, healthcare providers, and scientists continue their efforts to identify and find ways of treatment regarding the coronavirus infection (CoV), also known as Severe acute respiratory syndrome coronavirus 2. This is due to the fact that the COVID-19 infection is ongoing and there is a possibility of new infections or new waves of disease. In addition to this possibility, there is also the continuity of the COVID-19 infection (SARS-CoV-2). Researchers have a pressing need for adequate biomarkers that are associated with the progression of SARS-CoV-2 in order to stratify patients who are at high risk. Because the disease can spread so rapidly, patients need to be classified into risk groups as soon as possible after their diagnosis in order to make the most efficient use of available resources. In addition, new markers are required in order to identify patients who have a rapid progression of their disease, which can lead to death or a serious infection. It is essential to gain a comprehension of the viral pathogenetic mechanisms, as well as the cellular and organ damage, prior to the discovery of novel biomarkers. The clinical management, screening, and prevention of serious complications could all be improved with the use of reliable biomarkers. To effectively manage the COVID-19 pandemic, future prevention, prompt diagnosis, superior treatment, and precise detection are not only extremely important but also have the potential to assist in reducing the spread of the virus. According to the conventional medical consensus, biomarkers play a very important role in the prompt detection of the etiology, treatment, diagnosis, and prognosis of a disease. This work discusses emerging and known biomarkers for detecting SARS-CoV-2 diagnostics, prognosis, and treatment in order to assist the numerous innovations and investigations that are currently taking place.
References
Gautret P, Gray GC, Charrel RN, Odezulu NG, Al-Tawfiq JA, Zumla A, et al. Emerging viral respiratory tract infections—environmental risk factors and transmission. Lancet Infect Dis. 2014;14(11):1113-22.
Li F. Structure, function, and evolution of coronavirus spike proteins. Annu Rev Virol. 2016;3:237-61.
Chan JF, Lau SK, To KK, Cheng VC, Woo PC, Yuen KY. Middle East respiratory syndrome coronavirus: another zoonotic betacoronavirus causing SARS-like disease. Clin Microbiol Rev. 2015;28(2):465-522.
Thomas P, Baldwin C, Bissett B, Boden I, Gosselink R, Granger CL, et al. Physiotherapy management for COVID-19 in the acute hospital setting: clinical practice recommendations. J Physiother. 2020;66(2):73-82.
Mason RJ. Pathogenesis of COVID-19 from a cell biology perspective. Eur Respir J. 2020;55(4):2000607.
Badraoui R, Alrashedi MM, El-May MV, Bardakci F. Acute respiratory distress syndrome: a life-threatening associated complication of SARS-CoV-2 infection inducing COVID-19. J Biomol Struct Dyn. 2021;39(17):6842-6851.
Mogensen TH. Pathogen recognition and inflammatory signaling in innate immune defenses. Clin Microbiol Rev. 2009;22(2):240-73.
Costela-Ruiz VJ, Illescas-Montes R, Puerta-Puerta JM, Ruiz C, Melguizo-Rodríguez L. SARS-CoV-2 infection: The role of cytokines in COVID-19 disease. Cytokine Growth Fac Rev. 2020;54:62-75.
Polidoro RB, Hagan RS, de Santis Santiago R, Schmidt NW. Overview: Systemic Inflammatory Response Derived From Lung Injury Caused by SARS-CoV-2 Infection Explains Severe Outcomes in COVID-19. Front Immunol. 2020;11:1626.
Henry BM, De Oliveira MH, Benoit S, Plebani M, Lippi G. Hematologic, biochemical and immune biomarker abnormalities associated with severe illness and mortality in coronavirus disease 2019 (COVID-19): a meta-analysis. Clin Chem Lab Med (CCLM). 2020;58(7):1021-8.
Qin C, Zhou L, Hu Z, Zhang S, Yang S, Tao Y, et al. Dysregulation of immune response in patients with coronavirus 2019 (COVID-19) in Wuhan, China. Clin Infect Dis. 2020;71(15):762-8.
Bo XU, Fan CY, Wang AL, Zou YL, Yu YH, Cong HE, et al. Suppressed T cell-mediated immunity in patients with COVID-19: a clinical retrospective study in Wuhan, China. J Infect. 2020;81(1):e51-60.
Liu F, Xu A, Zhang Y, Xuan W, Yan T, Pan K, et al. Patients of COVID-19 may benefit from sustained lopinavir-combined regimen and the increase of eosinophil may predict the outcome of COVID-19 progression. Int J Infect Dis. 2020;95:183-91.
Long L, Zeng X, Zhang X, Xiao W, Guo E, Zhan W, et al. Short-term outcomes of COVID-19 and risk factors for progression. Eur Respir J. 2020;55(5):120376.
Tang N, Bai H, Chen X, Gong J, Li D, Sun Z. Anticoagulant treatment is associated with decreased mortality in severe coronavirus disease 2019 patients with coagulopathy. J Thromb Haemost. 2020;18(5):1094-9.
Yang X, Yu Y, Xu J, Shu H, Liu H, Wu Y, et al. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: a single-centered, retrospective, observational study. Lancet Respir Med. 2020;8(5):475-81.
Du RH, Liang LR, Yang CQ, Wang W, Cao TZ, Li M, et al. Predictors of mortality for patients with COVID-19 pneumonia caused by SARS-CoV-2: a prospective cohort study. Eur Respir J. 2020;56(3):2050524.
Bangash MN, Patel J, Parekh D. COVID-19 and the liver: little cause for concern. Lancet Gastroenterol Hepatol. 2020;5(6):529.
Xiang HX, Fei J, Xiang Y, Xu Z, Zheng L, Li XY, et al. Renal dysfunction and prognosis of COVID-19 patients: a hospital-based retrospective cohort study. BMC Infecti Dis. 2021;21(1):1-7.
Levi M, Scully M. How I treat disseminated intravascular coagulation. Blood. 2018;131(8):845-54.
Sardu C, Gambardella J, Morelli MB, Wang X, Marfella R, Santulli G. Hypertension, thrombosis, kidney failure, and diabetes: is COVID-19 an endothelial disease? A comprehensive evaluation of clinical and basic evidence. J Clin Med. 2020;9(5):1417.
Liao D, Zhou F, Luo L, Xu M, Wang H, Xia J, et al. Haematological characteristics and risk factors in the classification and prognosis evaluation of COVID-19: a retrospective cohort study. Lancet Haematol. 2020;7(9):e671-8.
Terpos E, Ntanasis‐Stathopoulos I, Elalamy I, Kastritis E, Sergentanis TN, Politou M, et al. Hematological findings and complications of COVID‐19. Am J Hematol. 2020;95(7):834-47.
Al-Kuraishy HM, Al-Gareeb AI, Al-Hussaniy HA, Al-Harcan NAH, Alexiou A, Batiha GE. Neutrophil Extracellular Traps (NETs) and Covid-19: A new frontiers for therapeutic modality. Int Immunopharmacol. 2022;104:108516.
Tan C, Huang Y, Shi F, Tan K, Ma Q, Chen Y, et al. C‐reactive protein correlates with computed tomographic findings and predicts severe COVID‐19 early. J Med Virol. 2020;92(7):856-62.
Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395(10223):497-506.
Meduri GU, Headley S, Kohler G, Stentz F, Tolley E, Umberger R, et al. Persistent elevation of inflammatory cytokines predicts a poor outcome in ARDS: plasma IL-1β and IL-6 levels are consistent and efficient predictors of outcome over time. Chest. 1995;107(4):1062-73.
Al-hussaniy H, Al-tameemi ZS. Methicillin-Resistant Staphylococcus aureus and New Delhi Metallo beta-lactamases-types of antibiotic resistance, methods of prevention. Med Pharm J. 2022;1(1):14-24
Fesseha H, Fesseha Y. Corona Virus Disease-2019 (COVID-19)-An Insight on Treatment Options and Prevention Protocol. J Comm Med Pub Health Rep. 2020;1(1):1.
Sanders JM, Monogue ML, Jodlowski TZ, Cutrell JB. Pharmacologic treatments for coronavirus disease 2019 (COVID-19): a review. JAMA. 2020;323(18):1824-36.
Al-hussaniy HA, Altalebi RR, Alburagheef A, Abdul-Amir AG. The Use of PCR for Respiratory Virus Detection on the Diagnosis and Treatment Decision of Respiratory Tract Infections in Iraq. J Pure Appl Microbiol. 2022;16(1):201-6.
ALZobaidy MA, AlbuRghaif AH, Alhasany HA, Naji MA. Angiotensin-Converting Enzyme Inhibitors May Increase Risk of Severe COVID-19 Infection. Ann Romanian Soc Cell Bio. 2021;25(6):17843-9.
Al-Juhaishi AM, Aziz ND. Safety and Efficacy of antiviral drugs against covid-19 infection: an updated systemic review. Med Pharm J. 2022;1(2):45-55.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2023 Journal of Clinical Trials and Experimental Investigations

This work is licensed under a Creative Commons Attribution 4.0 International License.
![]() The journal is licensed under a Attribution4.0 International (CC BY 4.0).
The journal is licensed under a Attribution4.0 International (CC BY 4.0).